The Basics, Details & Future of Stereotactic Treatments - Insights from a Medical Physicist
We had the opportunity to interview Masayori Ishikawa, PhD, a Senior Medical Physicist at the Hokkaido University Hospital and professor at the Faculty of Health Sciences, Hokkaido University. Dr. Ishikawa has been a Novalis-accredited auditor since 2015.
As an expert in the field of radiation therapy, Dr. Ishikawa shared his insights on the basics, details and future of stereotactic treatments as well as his experience working with the Novalis Certification program.
The Basics – SRS, SBRT and Radiotherapy
What is the difference between stereotactic radiosurgery (SRS) and stereotactic body radiosurgery (SBRT)? How does radiotherapy fit into this?
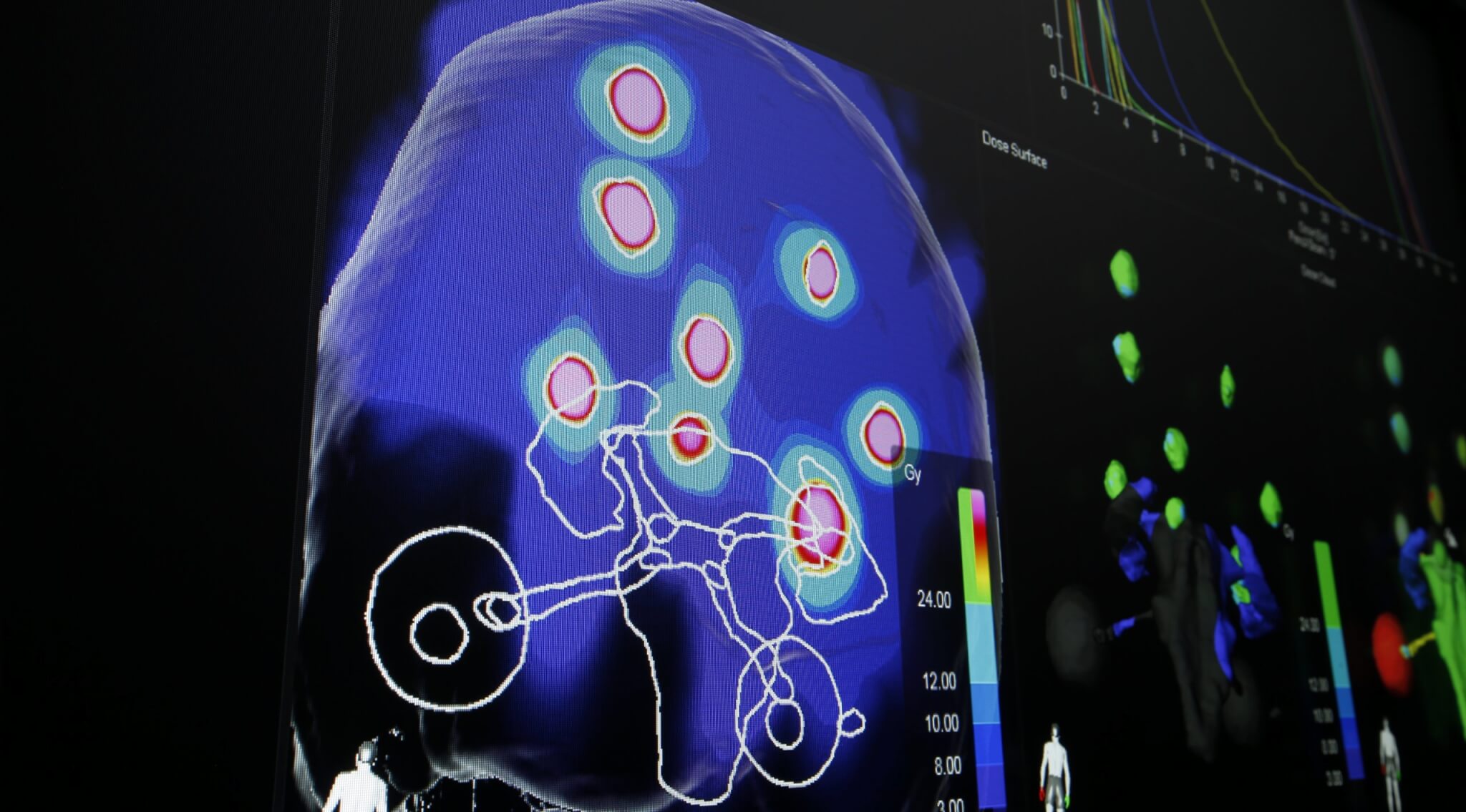
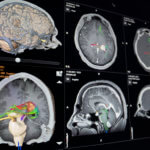
Dr. Ishikawa: « SRS primarily targets brain tumors, especially very small tumors (usually less than 1 cm). Stereotactic means « advanced position control. » During SRS administration, we are careful to avoid damaging healthy tissues in the brain as much as possible by performing pinpoint irradiation accurately with an error of 1 mm or less. In the past, SRS required fixing a metal frame to the skull with screws and then fixing the frame to the treatment couch to maintain precision. In addition, a high level of accuracy is required not only for position control but also for dose administration.
On the other hand, SBRT is also used to treat lung and liver tumors and, in recent years, for vertebral body tumors. The decisive difference from SRS is that there is movement inside the body. Since brain tumors have absolutely no possibility of moving within the skull, we can secure positional accuracy by firmly fixing the skull, but it is basically impossible to fix organs for SBRT. Therefore, there is a need for a technique that either restricts the movement of the organ or precisely irradiates according to movement.
In conventional radiation therapy, irradiations of more than 20 fractions are performed over a period of 1 to 2 months. With this technique, side effects decrease when higher fractionated irradiation is administered because healthy tissue can be recovered in lower dose irradiation. In comparison, by using the high-precision irradiation technology of SRS and SBRT, damage to normal tissue can be reduced in principle. Usually, treatments are performed using higher dose irradiation within a week with 1 to 4 irradiations. Since higher dose irradiation can reliably kill tumor cells, treatment outcomes are dramatically improved compared to conventional radiotherapy. »
How common are stereotactic radiosurgery, stereotactic body radiosurgery and radiotherapy? Are these types of treatments considered standard of care for the treatment of solid tumor cancers?
Dr. Ishikawa: « First, radiotherapy is practiced as a primary treatment for patients with unresectable tumors or those who cannot undergo surgery in many countries. This is largely because radiation therapy poses less of a physical burden than surgery or chemotherapy and is a treatment method that is easier to administer to the elderly.
As SRS requires advanced technology, it is used only at a limited number of facilities. However, in recent years, advances in treatment apparatuses and the spread of IGRT (image-guided radiation therapy) have improved patient set-up position accuracy, so many facilities can start offering SRS.
Since SBRT requires special equipment to restrict or follow the movement of the tumor, it is not practiced at as many facilities as SRS. But as equipment for performing SBRT is becoming increasingly commercially available, the number of facilities that have introduced these devices will also increase at the timing of equipment renewal. In particular, large hospitals will be able to implement these upgrades more easily.
Meanwhile, radiotherapy has become a first-line treatment, and it has been established as a standard treatment for many tumors. For example, radiotherapy is the standard treatment for nasopharyngeal cancer and oropharyngeal cancer. SRS/SRT is applied to brain metastasis and benign tumors such as vestibular schwannoma. Non-small cell lung cancer is a good indication for SBRT. In addition, by performing treatments in combination with other modalities, such as chemo-radiotherapy and postoperative radiation therapy, it is expected that the therapeutic effect can be enhanced and reduce the risk of recurrence. Breast-conserving therapy is a typical example of postoperative radiotherapy. »
As a medical physicist, what do you do during radiation therapy procedures?
Dr. Ishikawa: « The role of a medical physicist in radiotherapy is multifaceted. Some standard parts of the job include maintenance and management of the treatment apparatus and treatment planning systems, implementation of patient-specific quality assurance (QA) in high-accuracy treatments such as IMRT, VMAT, SRS/SRT/SBRT and maintenance of measurement devices for performing patient-specific QA.
In addition, treatment planning for IMRT/VMAT may be performed depending on the facility. In equipment maintenance and management, verification tests are performed according to their importance, such as every day, every week, every month, every half-year and every year.
Although it is not frequent, medical physicists also conduct acceptance and commissioning for newly installed treatment apparatuses. In daily treatment, medical physicists check the treatment status on a weekly basis, share information at conferences and respond to questions and requests from doctors and technicians as a medical physicist. »
The Details – Present-Day Accuracy to Future Real-Time Tumor Tracking
Are there any globally recognized standards for SRS and SBRT? If so, what are they?
Dr. Ishikawa: « Standards of cancer treatment in general, not limited to radiotherapy, vary from country to country. Differences in physique due to differences in race and differences in lifestyle habits play a major role. However, the results of multi-center clinical trials (RTOG, EORTC, TROG, JCOG, etc.) conducted in countries around the world are often used in guidelines established by each country.
In Japan, the NCCN (National Comprehensive Cancer Network) Clinical Practice Guidelines in Oncology (NCCN Guidelines) are frequently used to establish Japanese guidelines. For example, SRS is defined as the first-line treatment for metastatic brain tumors, and SBRT is defined as the first-line treatment for patients with inoperable early-stage lung cancer and patients that wish to forego surgery. »
When it comes to radiation therapy, maintaining high accuracy is key to ensuring a patient’s healthy tissue is spared. In your experience, what can a radiation oncology team do to improve accuracy during treatment?
Dr. Ishikawa: « Numerous studies have been conducted to define best practices for maintaining high-accuracy radiotherapy. In order to spare healthy, non-cancerous tissue, positional accuracy improvement and accurate dose delivery are important. Improving positional accuracy also includes patient fixation and the use of image-guidance.
On the whole, it is important to be aware of the latest equipment, have a clear understanding of the purpose of the equipment and be up to date on the related methods to use it. In case the facilities already have the latest equipment, the quality of treatment can be guaranteed by thoroughly maintaining and managing such devices. For accurate dose delivery, there are many items related to software, such as the beam modeling accuracy of treatment planning systems and quality control of treatment apparatuses. In particular, medical physicists are responsible for commissioning treatment planning systems and revalidating them when they are updated.
As a radiation oncology team, I think it is important to continue to collect information on the latest updates in the field by reading scientific papers and attending conferences. By collecting information from the standpoints of doctors, physicists, technologists, nurses and dosimetrists (in Japan, medical physicists also serve concurrently), we believe that treatment accuracy can be improved in many ways. »
You’ve conducted years of research on topics like radiotherapy and real-time tumor tracking. What exactly is real-time tumor tracking, and how can this type of technology shape the future of solid tumor treatment?
Dr. Ishikawa: « I personally lead the development of a next-generation real-time tumor tracking (RTRT) system, which is classified as a gated irradiation system, and have experience in manufacturing commercial devices and getting pharmaceutical approval. The current RTRT system requires fiducial marker implantation near the tumor for accurate tumor tracking, but the recent trend is markerless tracking by X-Ray fluoroscopy of solid tumors in combination with image recognition technology, such as AI.
In the case of gated-irradiation, irradiation is performed only when the moving tumor enters a predetermined range, so initially, there was a concern that the irradiation time would be longer. However, in recent years, this problem is being solved because high-dose-rate irradiation has become possible by using the flattening filter free (FFF) Beam. Furthermore, FLASH radiation therapy, which has been attracting attention in recent years, uses a very high dose rate of 40 Gy/sec or more, so more accurate tumor position recognition will be possible. »
The Overview – Standardization and Certification with Novalis
What is your role within the Novalis Certified program? Why did you choose to become involved with the Novalis organization?
Dr. Ishikawa: « My role in the Novalis Certified Program is as an auditor. In the Novalis Certified Program, we ask the medical institute to submit a document called “self-study pre-audit” to check how well the facility meets the standards required by the Novalis Certified Program. Ultimately, we aim to increase overall quality by encouraging the improvement of items that are not met with the standards, especially documents which are not clearly written. The auditor’s role is not only to check documents and facilities, but also to advise on the preparation of such documents.
When I was asked to be the auditor, there was no such certification program in Japan, so I was very interested in the system of the certification program. The auditor tasks are tough, but I learned a lot through the onsite visits. In particular, Dr. Timothy Solberg was present during my first onsite visit and taught me the points to be observed during the audit. Based on that experience, I can provide appropriate guidance to facilities undergoing the Novalis Certified Program. »
Why should a medical institution consider entering the Novalis certification program?
Dr. Ishikawa: « One of the many benefits of participating in the Novalis Certified Program is that medical institutes can learn how to guarantee consistent quality of comprehensive SRS/SRT/SBRT.
However, the program is not just about mastering the technical requirements of SRS/SRT/SBRT. It is also about improving the interest in training the entire staff, including physicians, as well as comprehensive safety management throughout the hospital organization, including outside the radiotherapy department. These are important points that can often be overlooking during daily practice.
From the standpoint of hospital management, participation in the Novalis Certified Program enables the treatment staff to fine tune their capabilities, and in parallel, provide high-quality, high-accuracy radiotherapy to patients. Ultimately, patients can be assured that if they or their family members or acquaintances are diagnosed with cancer, there is a facility that can consistently deliver high-quality care. »
As an expert in the field of radiation therapy, how do you anticipate the field will evolve in the next 5-10 years?
Dr. Ishikawa: « In recent years, an increasing number of facilities have introduced new modalities, such as proton therapy machines and MRI-equipped linear accelerators. As the field of radiation therapy continues to develop along with advances in science and technology, it is quite possible that even newer treatment devices will emerge. For example, linear accelerators combined with a PET scanner and MRI-equipped particle therapy machines are already under development for commercial use and may be ready to use for treatments in the next decade. However, these state-of-the-art devices are very expensive and are expected to be installed only in a limited number of facilities. It is likely that many facilities will retain conventional radiotherapy equipment for some time.
On the other hand, software is also evolving at an ever-increasing pace. In particular, AI (Artificial Intelligence) has developed remarkably and can perform tasks in image processing at a level that cannot be achieved by humans. Radiotherapy has evolved from reliable dose delivery to tumors by adding setup and internal margins to protect normal tissue by restricting and following movement in response to daily body shape changes and patient setups. In addition, adaptive radiotherapy, in which the treatment plan is modified to accommodate body shape changes, has been investigated to further increase tumor dose concentration. AI is very compatible with this adaptive radiotherapy, and the possibility to modify the treatment plan in a short time means that, just by performing daily CBCT scans, the appropriate treatment plan for that day’s patient set-up may be obtained in a matter of minutes.
The emergence of new technology may provide more accurate treatments, but in order to make full use of the performance of these devices, the quality of operators must also be improved. As mentioned above, the Novalis Certified Program not only provides quality management of equipment and technology, but also provides a comprehensive quality improvement program that includes the entire facility. »