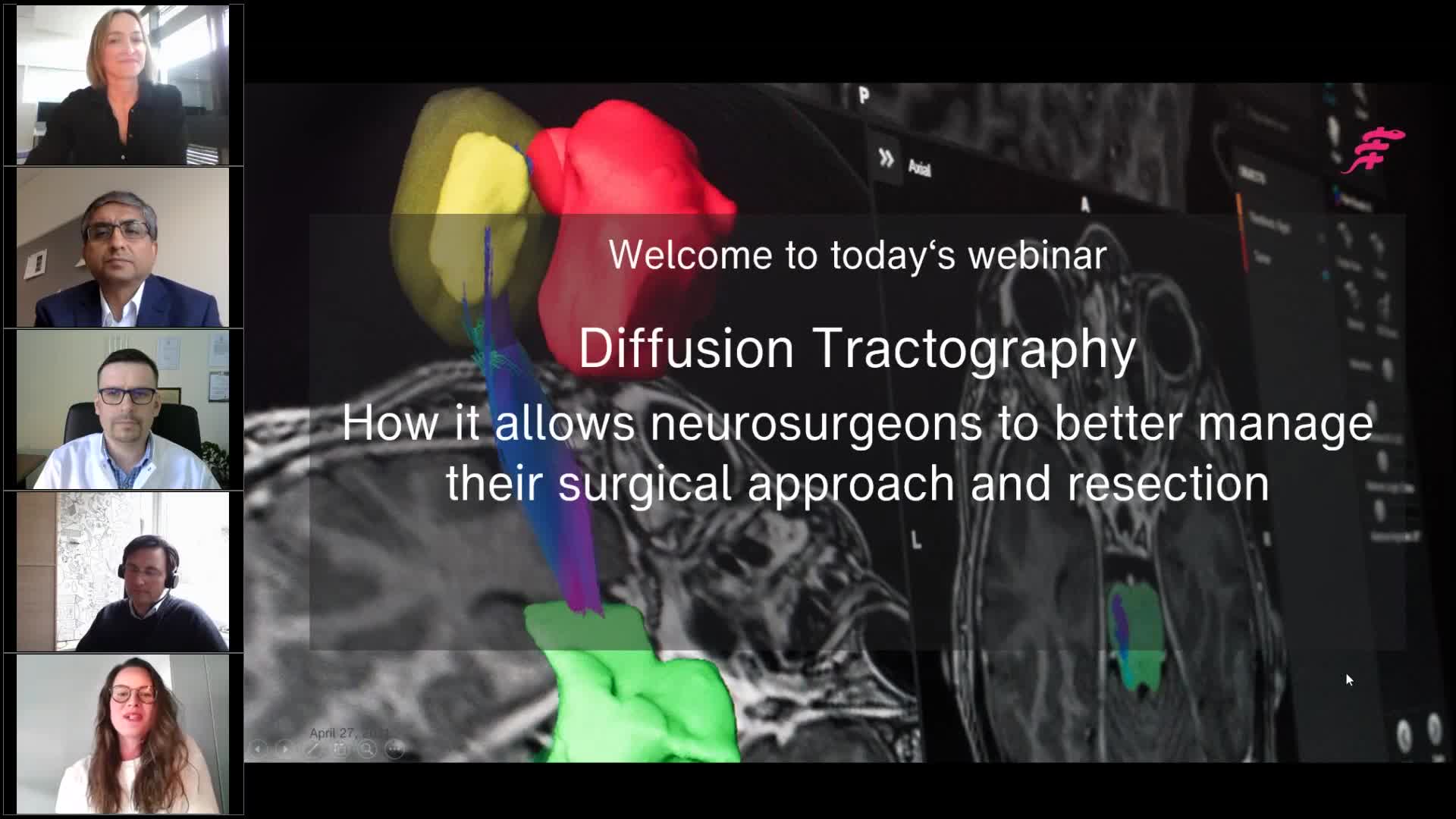
Webinar
Diffusion tractography - How it allows neurosurgeons to better manage their surgical approach and resection
描述
Brainlab invites you to join our live webinar, “Diffusion tractography – How it allows neurosurgeons to better manage their surgical approach and resection”, on April 27, 2021 at 4:00 PM CET.
The webinar will cover:
- Clinical cases where use of fibertracking completely changed the surgical plan
- How DTI facilitates neurosurgeons to better guide their surgical approach and resection
- How fibertracking can complement other intraoperative monitoring techniques
- Scope and limitations of this technique
We look forward to meeting you online!
Language | English
In case you can’t join the webinar, it will be recorded and shared afterward.
Participation is free of charge.
The views, information and opinions expressed during this presentation are the speaker’s own and do not necessarily represent those of Brainlab.
演讲人
Pawel Tabakow, MD
Wroclaw University Hospital, Wrocław, Poland
Einar Vik-Mo, MD
Oslo University Hospital, Rikshospitalet, Oslo, Norway
Video Transcript
Nadja: Afternoon, everyone, and welcome to today’s webinar on “Diffusion Tractography, How it Allows Neurosurgeons to Better Manage Their Surgical Approach and Resection.” First of all, thank you for taking the time to join us today. My name is Nadja Heindl and I’m clinical marketing manager for neurosurgery at Brainlab. It’s my pleasure to introduce our three speakers and experts for today’s session. We got Mr. Puneet Plaha from John Radcliffe Hospital in Oxford, England, Dr. Einar Vik-Mo from Oslo University Hospital in Norway, and Dr. Pawel Tabakow from Wroclaw University in Poland. These three are going to share their experience in using FiberTracking and will also happily answer your questions. Also with us in this webinar is my lovely colleague, Emese.
Emese: Hello and good afternoon and a very warm welcome also from my side. My name is Emese Csutak, I’m a marketing manager here at Brainlab. So, today’s webinar is going to last approximately one hour and we have a pretty packed program. And after the three presentations, there’s going to be some time for a little discussion and a Q&A session, so if you have any questions, you can go ahead and enter your questions into the chat box in your webinar interface at any time during this webinar. Those questions will be reviewed and can be discussed during the Q&A. And don’t worry in case you miss out on anything, this session is going to be recorded and shared with you afterwards.
Nadja: Thank you, Emese. So, before I hand over control to our first speaker, Mr. Puneet Plaha, let me briefly introduce him. Puneet Plaha is consultant, neuro-oncologist surgeon, and associate professor in Oxford with a specialist interest in brain tumor, especially endoscopic minimally invasive surgery and awake surgery. His area of interest is setting up novel clinical trials for brain tumors, advanced brain imaging for brain tumors, novel interoperative technology to minimize morbidity during brain tumor surgery, endoscopic minimally invasive brain tumor surgery, awake surgery, and immunotherapy for glioblastoma. He’s also a clinical investigator for the NIHR-funded FUTURE-GB trial and during our dry run, Puneet, you were telling us something about running, so what’s that got to do with the running?
Puneet: Nadja, it’s just a passion. I tell my trainees, “If you want to finish your training in Oxford, the key important point is to be a good runner and participate in half marathon once in your training.” So, that’s the message I give them.
Nadja: Very nice. Great. So, you’re a huge runner yourself, right?
Puneet: I am. Yes, thank you.
Nadja: Yeah. Okay, great, then, let’s go ahead and see your presentation, the stage is yours.
Puneet: You can see my screen? Sorry.
Nadja: Yes, we can see you.
Puneet: Okay, thank you for joining in. I’m going to talk about DTI, it’s a big topic, but I will focus on how it changes surgical approach for intrinsic tumors and give you a sort of bird’s eye view on our experience with this year. So, we know a lot about DTI, there’s a lot of publications, it’s a very valuable tool, there’s a lot of papers coming around on it, but we’ll focus on the one I’m highlighted in red, how does it help in the safest surgical approach and some examples. So, case number one, this is the gentleman with the left temporal glioblastoma. This is actually an intraoperative navigation view. And he presented with clumsiness and distorted vision. Now when you look at this scan, if I instinctively look at it, I sort of pointed here, that’s the sort of shortest route to get to it looking at this scan picture.
You could also take a view from below if you look at the long axis, but you’ll pretty much choose an approach within this area. You then put them to a tractography scan, you get these very colorful 3D reconstruction pictures, the tumors in red. As expected, the tumor is encased by the ILF, the IFOF in green, you get the cortical spinal tract immediately. Now when you look at this, suddenly your perspective changes because what was planned as an approach now with…this will be the shortest access route, now with your tractography, I think you have a revised approach. You change your approach because the green in the IFOF and the sort of the optic radiation, you have a bit of…sorry, the IFOF is in green, and then you have a bit of the ILF and optic radiation coming in your way on the shortest route, so you change the trajectory going slightly higher up.
This is the revived approach. This is the 3D picture reconstruction, so the surgeon’s view is what you see in the middle, that’s your access corridor to try and avoid fiber tracts, and that’s just to the superior part or the posterior part of the middle temporal gyrus. So, this patient goes through an awake, I’ve just put an intraoperative picture, we try and follow this trajectory through here, get the negative cortical map at four milliamps and choose the other entry point to resect the tumor. As you can see, I don’t have a post-operative DTI but I’ll show you other cases with postoperative DTI, but I’ve just shown the representative post-operative MRI scan to show that actually, the route pretty much matches what we had planned preoperatively to avoid the tracts.
As you would expect, given your tract, there is some sort of impairment you will always see after surgery and that improved significantly over a couple of weeks’ time. Now, this is a second tumor. Now, again, this is a tumor in the left posterior medial temporal The difference here is this tumor is medial to the HM or the ventricle, the previous one was lateral to it. Now, this lady presented with confusion, short-term memory impairment, and word-finding difficulty. Looking at tractography, as expected, you have the tracks, the sagittal stratum lateral, this is the tumor growing medial to it. Now, you could possibly say that’s the shortest route to it and the direct route, it’s going through a dominant lobe. But even if you take a much lower route through here, you’re actually going through the visual word form area up to this tumor, not really optimal.
So, what do we do? So, we did an endoscopic approach, and this is published. We did the occipital posterior ipsilateral interhemispheric with the tumor side down to resect the tumor, you can see the post-operative scans. for various reasons, she couldn’t go to an awake either. And as you can see here, the postoperative DTI scan shows that the tracts are intact, cognitive impairment persisted, she didn’t have a visual field defect, and there’s some residual tumor on the lateral aspect there, but a different approach from what you would expect till you saw that fractography images. Now, this is a…I’m just going to show this. Another interesting case, where I’m going to show you two cases with the same tumor in same location but tractography is totally different.
Thalamic GBM, thalamic pilocytic astrocytoma, presented as an emergency, you see the tumor in the thalamus, the cortical spinal tract is medial to it. This is a good approach, patient has this approach of an emergency, we did endoscope-assisted and come laterally and resect. You see the post-operative DTI scan, the cortical spinal tract is intact, there’s a small residual tumor. Now, that’s just the endoscopic approach. I’ll show you now, this is a patient, he was referred a few weeks after the previous one, exactly the same, so we look at it and said, “We’ll do exactly the same approach as on the previous case.” We now get a tractography scan and interestingly, the cortical spinal tract is lateral to the tumor. So, what it means is you cannot then come to the approach as you had done in the previous case.
So, same tumor, same direction, same location, but the tracts differ and your approach differs. How did we get to this? We then did an endoscopic left frontal, we enhanced for the cortical. This is just the endoscopic approach, endoscope-assisted, you can see the endoscopy enter the ventricle. You make your tract with the surgical patties, the thalamus stays with corticotomy, you enter there to resect the tumor. Now, the point here is tractography is invaluable in changing your perspective and direction of approach. I’m going to move this forward with the video but that’s just resecting the tumor. Now, go back to the temporal lobe again, slightly there’s a difference, non-enhancing glioma in a young gentleman presented visual symptoms. You can see this tumor, we had some functional imaging done for him to map the areas, and this is DTI.
Now, you see a mixture of things here, you have the IFOF lateral, you have the ILF, optic radiation, the awkward turning around, there are multiple tracts. How do you get to this? The shortest route or you do the safest route? The shortest route is not the optimal because you may damage the tracts but you use cortical tractography to choose the safest route to get to this tumor. And this is the result, it’s done awake, and as expected, we have errors during surgery with IFOF, with ILF anomia, and ultimately vision, but they improve significantly. The post-operative scan, this is the post-operative DTI scan, you can actually see fiber tracts here showing that they’re intact following the surgery, the optic radiation, the ILF, and the IFOF here. So, just to show that tractography maximizes resection, as expected, we didn’t achieve a complete resection but a very good resection on this case but minimize the deficit.
Now, this is again a low-grade glioma, slightly different in the sense of the tumor starts from…it’s a mesial parahippocampal gyrus non-enhancing glioma, where you have the non-enhancing tumor here and the young lady presenting with word-finding difficulty. Look at the slide in between, we have the preoperative DTI scan. If you look at it from a lateral perspective, the sagittal picture, you will see the tracts are encasing the tumor pretty much on the lateral aspect and on the superior aspects. Your only real window is actually if you come low down at the posterior and then go through the visual word form area here, possibly an approach but the patient will have difficulty with reading and writing, or you take a slightly different approach. So, we approach this through an endoscopic approach coming from the front, anterior medial to the tracts that I’ve shown here. And this is the post-operative DTI on the right side, taking medial to the tracts.
You will take away, with this approach, the anterior part of the ILF, the patient does have temporary anomia but they actually do recover from it very well. In this case, just to show the post-operative DTI scans, the patient did have difficulty with speech as one would expect but did improve about a month following surgery. This is my last case. Now, I sort of stuck to temporal lobe just to show you a flavor of different temporal lobe tumors and what different modalities one can use. This gentleman presented with headaches, this is a tumor high upon the sagittal stratum. You can see tracts on top and lateral to it and there’s tracts of the sagittal stratum with the IFOF and the optic radiation. The red one shows a direct approach to the tumor, you would go to the middle temporal gyrus, but you likely will damage the tracts. We took a different approach here with the endoscope-assisted, we come low along with inferior temporal gyrus and then move up underneath the optic radiation.
What I mean by that is, if you see this picture in the bottom right, you see the preoperative DTI showing the optic radiation on the superior lateral aspect of the tumor, and this is the post-operative one showing the optic radiation intact. The top is the visual field defects pre and post, if anything, the post one is better because some of the mass has been taken away. But we’ve done a low endoscope-assisted approach that I’ve shown in this cartoon here, coming low along the temporal lobe, underneath the optic radiation, and then going up. This is just a video of that case, this is along the temporal lobe, you’re looking up using 5-ALA fluorescence, you can see the fluorescence of the tumor. You take a tumor biopsy, and then you resect with the diffuser, and that’s just sort of finishing of the resection with the endoscope, and that’s looking for further 5-ALA residue as you can see in the corner to resect that aspect of the tumor as well.
So, we’ve now moved this forward with our colleagues in Imperial with a trial called FUTURE-GB which are looking at both the value of diffusion tractography and ultrasound in improving resection and more importantly quality of life of the patient. It’s NIHR-funded, we open the trial in November, it’s likely to run for the next five years, and I think it will give valuable answers around DTI. So, that’s my take-home message, I think from a surgical perspective, I’ve shown you a small cohort of seven patients but I can show you a larger series which we have used DTI for every case, it’s invaluable surgical planning. Also, postoperative DTI is extremely helpful from a learning perspective from the surgeons, if you plan something before surgery, you want a post-op to see how you’ve done. And then, obviously, FUTURE-GB will give us more answers on quality of life in terms of resection and morbidity. Thank you very much.
Nadja: Great, thank you very much for your presentation. I actually would like to move on to introducing the next speaker already since we have the Q&A session at the end. So, Einar will be our next speaker. Now, Dr. Einar Vik-Mo is heading the surgical neuro-oncology section at the Oslo University Hospital in Norway, which is one of the largest in Scandinavia. Einar has a Ph.D. training in tumor cell biology and immunotherapy and is a principal investigator on several early phase studies on innovative therapies. His main surgical focus is on malignant brain tumors and intraoperative imaging and monitoring. And Einar, I think you also have a little story to tell us, right?
Dr. Vik-Mo: Now you can hear me? Yeah.
Nadja: There we go.
Dr. Vik-Mo: So, neurosurgery is scary and we talked about some of these are things that can happen that are scary. So, my favorite pastime is scuba diving, so once I was diving, I actually had a monkfish that stuck on to my knee. So, getting out of trouble is a useful thing to know both in neurosurgery and when you do diving. So, that’s kind of my fun fact. So, fantastic talk by Puneet showing the versatility of using DTI. I’m going to show how we are doing this in Oslo. Just a short disclaimer and disclosures. Just very briefly, Oslo is the capital of Norway, we’re covering about half of the Norwegian population, and we have about 600 tumor surgeries here. Just so you know where you have me, I am a strong believer in the careful workup and that is essential, and that fMRI and DTI, they’re only hypothesis-generating.
So, I think it’s really important to understand that this will allow you to set up your surgical strategy and then you can use this to actually improve and really pinpoint your interoperative monitoring. I’m a strong believer in individualized treatments and this is what Puneet also has been showing, and I think it’s important to include patients in the decision making and to figure out where on the onco-functional balance, you get an adequate treatment strategy. We can’t do any neurosurgery without good imaging and I think it’s important that we take this, not only DTI, but put it into a broader context, and I would say that we need to figure out tumor anatomy. We mostly use preoperative with gradient-echo for the T1 contrast enhancement so we can see the vessels, we look at the invasion patterns specifically also on FLAIR, we look at the intratumoral heterogeneity, especially looking for the more malignant parts and making sure that we really get a good resection of those.
And then it’s to identify these functional important parts within close vicinity of the brain and we tend to use DTI much more than we use fMRI but we use both. And then we come to the neurosurgical armamentarium, and it’s a range of tools you can use and there are tools. Of course, we use neuronavigation, we combine this with ultrasound, MRI, fluorescence imaging, awake craniotomies, neurophysiological monitoring by motor evoked potential which I think have changed glioma surgery immensely, and also bipolar cortical and subcortical stimulation. But this meeting is about tractography, right? And we need something that gives us more of these really nice pictures, and I’m trying to do this more of a hands-on approach. I think it’s very important when you start doing this is that you accept that the DTI is very…it’s very artifact sensitive and you tend to get these distortions along the long axis of acquisition.
And for most of the cases, preoperatively, this doesn’t really matter much, it has some effects but only slightly. But if you’re looking at intraoperative or postoperative images where you have introduced air into the cranium, then you can have quite a lot of distortion. So, distortion correction is extremely important. I think this is…mapping out the different classical subtract is something that the neurosurgeon has to do themselves and in planning, so we often ask, “So what parameters do you use?” So, this is my basic setup. There are ways of mapping out these, you can do this by the tools that are in the system and whether or not you use a dynamic tool or regions of interest doesn’t really matter much, but you need to know what you’re looking for. And I would say that most people when they start out, do the cortical spinal tract, do the arcuate fasciculus, then you could start mapping it to all of these other more complex fasciculi as you go along.
One way of starting out is, of course, to do the cortical spinal tract, and here you have the hand knob region as defined within the software and you get this very nice tract. You can, of course, split these and you can look at the more of the tracts that go to the foot…or the hand, but what you immediately can see is that the lateral part of the precentral gyrus doesn’t really give rise to a lot of fibers. That is, of course, due to the curvation of these fibers within this region and it is actually quite difficult to map out the lateral part of the cortical spinal tract. For neurosurgeons, we know that there is a bilateral innervation of the face and swallowing but still, it’s important to understand what you’re mapping out and when you start out at this, be aware that this is…when you map out this, you get the medial part. For the language-associated tracts, this is a patient operated originally almost 10 years ago for what we now would call an oligodendroglioma.
And within this tumor, you see there’s a region here of reduced diffusion, suggestive of higher cellular content that actually has a higher oxygen consumption, which you can see in the fMRI, and there’s also incremental perfusion. This is a right-handed patient, and you can see that actually, she activates on both sides when it comes to reading. So, in here, you can map out the cortical spinal tract, the arcuate fasciculus, the IFOF, and the uncinate fasciculus. And then you take this in and you see that…you combine it with the fMRI and you see that your arcuate fasciculus actually maps quite well into the fMRI and you see that the cortical spinal tracts get pretty close to this region. Then you keep on simulating the surgery preoperatively and going through this, you see, “Yeah, this is the region that I’m most interested in having a resection,” and you can see that it’s pretty close to the arcuate fasciculus, not so far from the cortical spinal tract.
But if I’m doing this careful resection, not going into the complete FLAIR volume, then, of course, I’m pretty safe even with IFOF, but going further down doing more aggressive resection, we need to monitor this more carefully. So, before going to surgery, you know which part of the tumor you need to map specifically and looking at more of a cortical spinal tract infection, looking at expressive dysphasia, or more of linguistic dysphasia. So, getting into more of a concrete case, this is a previously healthy man, he had removed a malignant melanoma previously, and then he was admitted to acute care. He had a central facial palsy and right-hand clumsiness, then you can see this contrast-enhancing lesion pretty close to the precentral gyrus. You can see that it has these islands of contrast-enhancing tissue medial to the larger lesion.
You can see that on spectroscopy, it’s more of a…the most malignant part is actually on the medial side of this necrotic lesion, of potential necrotic lesion. And looking at FLAIR and also the T1, you can see that the pathological signals actually moves into the corpus callosum and down long to now what we think would be the cortical spinal tracts. So, if you take this into your system and you analyze it and you just put in an FA value of, say, 0.3, you end up with this kind of setting, you see that you have a split in the cortical spinal tract because it’s affected by the FLAIR signal. Then you can manipulate this, you go lower on the FA value, you have a shorter length and you allow a higher degree of angulation, you can actually start dissecting fibers within this FLAIR signal.
And pushing the envelope even further here, you can see this tumor is partially pushing the cortical spinal tract medially, so the contrast-enhancing, the malignant part is more pushing the cortical spinal tract while it’s clearly invading through it. So, having a medial view, you can look into the cortical spinal tract for the medial side and you can see that the contrast-enhancing tumor is lateral, so you can actually approach this through a lateral approach. You can then map out the arcuate fasciculus, you lower the threshold, you get more fibers in, and you get a clearer definition that you’re pretty close to the arcuate fasciculus in the inferior side of the lesion. So, going into the surgery, you can see that the IFOF is pretty far off, the arcuate fasciculus is pretty close, you need to have the patient awake, and you can actually push it quite far to the medial side, so you have the potential to take away most of this contrast-enhancing tumor.
We go talk to the patient and we need to balance this onco-functional strategy for him. Definitely, he cannot have acquired aphasia, he needs to be mobilized, otherwise, his survival would definitely be shorter, he needs to be walking. He has already affected speech and eating but he is content with the way it is and he is willing to put his hand on the line. Because on the other hand, you need to take out as much of the tumor as possible and we know that the contrast-enhancing part for this patient is quite rapidly growing and we know that if he’s going to have usage of other treatments, we need to pre-condition for those. So, this is just to show you the intraoperative image, this is the Sylvian fissure, we map out the precentral, we get the original speech arrest. And in the interest of time, what I’ll do is I’ll move this ahead.
We do map monitoring, we identify the tumor, we resect the fluorescent part, and we get to the point where we do intraoperative subcortical simulation bipolar, we get to speech arrest, so we’ve identified the arcuate fasciculus. And on the medial side, we get pretty close into a three milliamps with an awake patient. And at this point, we put the patient back to sleep, and we do the intraoperative MRI. The set that we have in Oslo is a 3 Tesla Philips system, we take the patient from the operating table, roll him into the MRI, and this is the intraoperative image. And I was kind of under the impression that I’ve taken out all the malignant tumor, but here you see there’s the island of this malignant tumor that’s left and between me, “That isn’t part of normal tissue.” And then I was thinking, “Okay, maybe it is,” so there are functional fibers in this region. We have the intraoperative diffusion series, which is highly affected, as you can see, and you need to correct it.
This region, which is on top, is highly affected. If we go back and forth between the uncorrect and corrected version, you can see that on the cross of the image, there is distortion in the image, but in the angle from top to bottom, it’s not as much distortion. And then we can do the intraoperative tractography, we see that the contrast in these lesions which are marked red from the medial side, there are fibers on the medial side, and from the lateral side, they are approachable. So, we go back in and we identify these. So, postoperative, the patient started out with a paralysis in the arm and we were thinking, “Okay, maybe we’ve gone too far.” We do the post-operative tractography, this is the resection cavity, and if you just add in standard parameters, you see there’s a hole in the cortical spinal tract.
Of course, this is disappointing but if you then tweak the fiber, you allow for a higher selectivity, you can see that we’ve been pretty close to the fibers but they are intact. And you see this on the axial and also on the coronal section, so the fibers are there. So, for the medial side, you see they are still intact. So, we see that we’ve been able to take out all the contrast-enhancing tumors being pushed all the way to the cortical spinal tract. And this displays also the sensitivity of the motor evoked potentials are may be oversensitive when you have the patient awake, but you get a higher sensitivity and specificity when you have the patient asleep. Another case, this is a patient with a lower parietal lobe contrast-enhancing tumor, and you can see that this would be pretty close to the arcuate fasciculus, which you see here.
We did weight craniotomy, did the resection, during the resection, we identified a combined dysphasia but when the patient woke up afterwards, she had more of a high degree of dysphasia and we saw this small region and then we worried maybe we’ve gotten small ischemic lesion here. We then did the tractography and we can see that the tracts actually are anterior to the ischemic lesion and they are passing through, so, again, we think this is a lesion that will improve. So, this is kind of the take-home message. The DTI supports pre, intra, and post-operative decision-making. It’s a skill that must be practiced and trained, and I think it’s useful for neurosurgeons to do that on their own and work with it. And it can help you build hypotheses for individualized treatment decisions but we still need monitoring, it doesn’t really…it doesn’t substitute the way craniotomy is monitored. And great thanks to the team because this is a team effort and to the patients that really allowed me to share their data.
Nadja: Great, wonderful. Thank you so much for also this very insightful presentation, Einar. So, I would actually like to move on to our last presentation of this webinar today, which is brought to us by Dr. Pawel Tabakow. Pawel became a consultant in neurosurgery in 2009. Since 2017, he is heading the neurosurgical department of Wroclaw University Hospital in Poland, and his scientific and clinical interests are strategies in triggering regeneration of the lesion fibers in the spinal cord transplantation of olfactory ensheathing cells and other spinal cord reconstruction techniques, functional neurosurgery including deep brain and spinal cord stimulation, epilepsy surgery, and application of computer-brain interfaces, neuro-oncology novel therapies for treatment of malignant gliomas, and also image-guided neurosurgery and neuro-endoscopy. And also, Pawel has a very interesting story of what happened to him on a holiday in a very tiny village in Sardinia.
Dr. Tabakow: Oh, well, actually, my team did a very interesting operation on a patient with spinal cord transection at the thoracic level, it was published in 2014, and there was much media interest around this, so I wanted to have some interesting holiday. A couple of years later, I went with my family to Sardinia and we were just thinking that nobody knows us, at that part of the world, nobody knows me or what I’m doing because no surgeon. At some very nice sunny day, when I was paying my dinner, the waitress just…after reading my name, recognize that I was the neurosurgeon behind the spinal cord reconstruction project. Finally, she said that she’s studying biomedical engineering and listen through during the lecture about our work. So, the conclusion, in a place where it has 300 inhabitants, we still can be recognized.
Nadja: Maybe she can visit you one day.
Dr. Tabakow: Yes, I invited her.
Nadja: Great, that’s fantastic. We’re looking forward to your presentation.
Dr. Tabakow: Okay, my presentation will be practical in terms of showing some tips and tricks how I use FiberTracking-based navigation, mainly during brain tumor surgery. I will start with a sentence from a famous neuroanatomist and psychiatrist, Bernard von Gudden, who said in the 19th century that anatomy is most important, “First anatomy, then physiology, without knowing the anatomy, we cannot go to physiology.” How we can learn anatomy? Of course, by learning new radiology contemporary scans showing brain topography and function but also during white matter dissection courses, if we talk about fiber tracts, for example, having formalin-fixed brains, you can do very nice white matter dissection like shown on this example where we see the horizontal and vertical part of the superior longitudinal bundle, also on the right side, we can see going further, the optic radiation, the arcuate fasciculus, the Meyer’s loop, the anterior commissure.
But the problem is that during the surgery, when you do the surgery, you can hardly find those tracts even you know from that white matter dissection course what is the topographic relation to that and this is why the usage of FiberTracking is very, very practical and important. So, in our department, we are using 3 Tesla imager and a very simple protocol for DTI, we also have a synthetic one using more directions of diffusion. And in general, you can build the fiber tracts manually or automatically. When you go to the manual form, most important is to use either the probabilistic approach or the deterministic. Probabilistic means that if you put a region of interest, for example, on FA colormaps, the system will build all the fibers that run to that area, to that ROI.
And in the deterministic approach, you’re using two ROIs and the system is showing the fibers that connect those two areas. Deterministic means more specific than probabilistic but probabilistic is easier but less specific. So, what we can build manually? I listed here, pyramidal tract, SLF, fasciculus arcuatus as a part of this fasciculus, optic radiation, fasciculus uncinatus, cingulum bundle, and many tracts used for DBS surgery. When it comes to the automatic, to the templates, they are based on automatically segmented areas of interest, and now we can use available templates for the pyramidal tract including the hand knob for fasciculus arcuatus, connecting the broken ventricles area, as well as for the optic radiation when the ROIs connects the lateral geniculate body and occipital lobe.
And generally, the general principle our radiologists are teaching us that we should use the FA map for reconstructions of the tracts and we have to put our ROI perpendicular to the orientation of tracts. But you can also build the tracts using T1-MPRAGE images, there’s a very nice paper about this from 2019 talking about language tractography, building of tracts involved in language and memory function, and this is a completely different way of how they used the ROI placement. But I’ll show my way. I’ll start, first of all, with the superior longitudinal fasciculus. Fasciculus is known for the initiation and regulation of motor behavior as well as language function, if we talk about fasciculus arcuatus. I start with a probabilistic approach and I’m looking at the coronal scan literal to the pyramidal tract, I’m establishing the first ROI here, and then I have a view like this.
I build the horizontal part of the SLF, sometimes if I’m very strict, I will have part of the vertical one but if I’m not satisfied with this and want to go deterministically, I will use the axial scan to establish a second ROI and there is a very nice interactive option, a white circle you can move in different directions and you can add or remove tracts. In this form, you can enrich your tractography. And in comparison, we see the automatic template for fasciculus arcuatus that is a bit thinner than the one I showed before. This is the same patient with left temporal low-grade glioma that I will discuss as my main case. And now how I build optic radiation before you had that nice anatomical segmentation map from Brainlab, we use that probabilistic approach on FA maps and this time, first I went to actual scans lateral to [inaudible 00:38:17] and establish the ROI on fMRI scans and I built two tracts, optic radiation and inferior front occipital fasciculus because they are very close to each other.
It was not easy to distinguish both tracts but for this surgery, both tracts were useful. And now we have at this moment also the template that connects the lateral geniculate body with the automatically segmented visual cortex, we see the Meyer’s loop and other portions of optic radiation, very nice in this case. Another patient with bleeding from cavernoma and in this case, I show a manually built fasciculus arcuatus that is medial to that pathology, and medially and posteriorly, we see the pyramidal tract from the automatic template. Why are you still on the automatic template? Because we can have the head knob option with a yellow arrow is pointing to the areas of fibers that go to the hand and face. SNr set is very, very difficult to go more laterally with photography, with deterministic photography, and the red arrows showing the fibers going to the trunk and lower extremities.
And in DBS surgery, we have established our own way of building functionally relevant tracts using our own templates. For example, if we want to operate a patient with Parkinson’s disease and we go with the electrode to the subthalamic nucleus shown in green here, first of all, we used anatomical segmentation, we were finding STN, then we are segmenting the limbic cortex shown in light blue and orange. This is the ventral medial part of the frontal lobe, then we are creating the hyperdirect pathway, the motor one from the premotor cortex to STN, and finally, we are connecting the dorsal lateral associative cortex with STN. So, in that form, we are doing functional segmentation of STN and this is the parcellation of the cortex of the frontal lobe we did by ourselves.
So, the light blue is the precentral gyrus, the green is the premotor cortex, the pink and violet we have the lateral association cortex, while the orange part shows the limbic system. So, you can create additional templates or functional relevant tracts and during DBS surgery, we are using the tractography-based target localization. For example, in a patient with Parkinson’s disease, we want to enter with the electrodes, the area where the hyperdirect pathways crossing the STN, so it’s the superolateral part of STN. And if you have a patient with obsessive-compulsive disorder, we’ll look at the area where the limbic tracts cross the anterior medial portion of STN, as shown here. Or if we want to treat a patient with essential tremor, we use the VIM, the ventral intermediate nucleus as a target, but also we have to build the dentato-rubro-thalamic tract shown in green, but that’s all for DBS.
Our main topic today is oncology, brain tumor surgery, so I go to my case presentation. So, we operated on a patient, a 36-year-old female suffering from temporal lobe epilepsy, abscess attacks, biopsy shows that it was a fibrillary astrocytoma. Initially, the patient has no neurological impairment. So, you see on FLAIR sequences, the area of low-rate glioma, the fasciculus arcuatus showed each horizontal vertical part, we see that the vertical part is very close to the posterior border of the tumor. Then, we build the inferior front of occipital fasciculus that is above the tumor, its very close to the superior temporal gyrus. And then we can see on this slide, the optic radiation that is superomedially to the posterior border of the tumor. And our 3D reconstruction in the same patient is showing the manual way and the automatic way of how we build the fasciculus arcuatus and optic radiation and inferior front occipital fasciculus, we see the 3D reconstruction of the tumor.
I would say that the manual way seems to be much more practical, may be less sensitive, but I’m showing now during that surgery on a very nice example of how I use the manual way. But if we look on the automatic template of fasciculus arcuatus, we will see that pink fiber that go to the Wernicke area, we cannot see in the manual one. So, you can combine both manual and automatic reconstruction of fasciculus arcuatus to have better results of cortical mapping. Another 3D view of the manual reconstruction versus automatic. Please note that the automatic template for optic radiation reconstruction showing also fibers that are part of the inferior fronto-occipital lobe, so we can erase those fibers too. Entering the surgery, we did awake craniotomy using controlled sedation with dexmedetomidine.
After scalp anesthesia and opening up the bone flap, we use the navigation tool to delineate on the cortical borders of the tumor, the anterior and posterior borders. But as [inaudible 00:43:44] saying, “First see the brain, then see the tumor,” so we use our microscope navigational tool, showing the focus of the microscope as the blue light, and we were looking at two specific orange endings from the fasciculus arcuatus. And let’s have a look at these two orange endings, I did on purpose now intraoperatively, I set the focus of the microscope not to focus ideally. You see those two and four dots, they don’t come as one, I did it because I wanted to see how the tracts from fasciculus arcuatus are superimposed on the brain cortex.
In that moment, we use bipolar stimulation and we started with finding the M1 cortex, we called that the evoked disartria, and this is a confirmation of the primary motor cortex from the face, face muscles, so, Brodmann number four. Then we went anteriorly to the area suspected to be Broca’s area, the patient was counting from 1 to 10 and we’re using bipolar stimulation from 2 to 8 milliamps 60-hertz, we’re trying the pulses system, and we confirm the Broca localization in front of poll number four. Then we went posteriorly, you see now the vertical portion of the fasciculus arcuatus at the level of angular gyrus, we confirm here by cortical stimulation amnestic aphasia here, the patient was not able to name different nouns, being shown different pictures. We also map the Wernicke area, so Brodmann area number 41. And now we will see a very short film, I guess. This film is showing the augmented reality 3D view of my tumor, the vein is in yellow, I use it just to see if there’s any distortion of the navigation.
We can use very easy intraoperatively correction of this navigation as far as we don’t have brain shift, very extreme brain shift. So, this is the way we monitor, the patient is speaking continuously, in that moment, being shown by our neurophysiologist pictures, he’s fully awake. And this is how we map the patient but now we have to go to the surgery. So, what do we have? We have a 3D view of the tumor, we see that part of the tumor invades the superior temporal gyrus, the eloquent area we map here, so we have to start the surgery from the non-eloquent part of the tumor. And with the superior technical resection and the use of chooser, I started the resection and we turn from mapping to continuous monitoring of the patient during different speech tasks.
And now we’ll see another short film by the end of the surgery, this is how we focus the microscope to the medial and superior medial part of the tumor and we found the inferior fronto-occipital fasciculus and somewhere here, it was supposed to…we also were expecting the inferior retinal fasciculus that is involved in nonverbal memory, face recognition, recognition of known objects. At this moment, we were showing the patient pictures of known persons, of Polish presidents, the patient was naming the presidents. We use at the same time the monopolar rabbit suction using the [inaudible 00:47:17] rule, that is saying that if you have a positive response at one milliampere, so we are one millimeter aside from the important tract. When we went posteriorly with that monopolar probe, we were able to cause transit features from fasciculus arcuatus, so we knew at what stage to stop the surgery by performing as maximally safe resection as possible.
And now, finally, even though we had some brain shifts, we were able to map not only fasciculus arcuatus but also the inferior fronto-occipital fasciculus, the optic radiation too, and it was very important that we could…by using the tractography, we could direct our navigated microscope to the exact area with the exact tracts we are expecting and we could use the exact test, either speech test or memory test or visual field evaluation. So, in conclusion, we find the FiberTracking technique very helpful both in the process of surgery planning, but also during the surgery, glioma surgery, DBS, and epilepsy surgery. Nowadays, we can use the probabilistic approach as well as the deterministic approach.
The newest approach like global tractography still a future, both manual and automatic visualization can be used. We cannot resign from manual and just go to the automatic one, we have to learn about tractography and how to build the tracts by ourselves. Intraoperatively, especially at the beginning of the surgery, that FiberTracking navigation enables the early identification of eloquent cortex areas and it improves cortical mapping. And during the surgery, the subcortical intraoperative tractography enables quicker identification of functionally relevant white matter tracts and the desired fiber bundle and quickens the time of choice of appropriate intraoperative tests for neural monitoring. Thank you for the attention. So, I have also attached a very interesting film from the same surgery.
You’ll see the patient before and after surgery with his MRI scan before and after, with translation from Polish to English. If we have time, we can show but I will leave now to the organizers. Thank you.
Nadja: Pawel, thank you very much for this presentation. Since we have 10 minutes, depends on how long the movie is…
Dr. Tabakow: Actually, I can speed up the movie, it’s part of our course of awake craniotomy, just to have a look. Once again, at the preoperative MRI.
Dr. Tabakow: Then we’ll have a quick look on patient positioning navigation. We use very often large openings in order to map as much brain as possible. We use the scalp block method system. This is a patient during surgery with constant contact with the neurologists and psychologists and for mapping of the Broca’s area, the patient is counting and the patient is naming pictures for tests of the Wernicke area and for the test of non-verbal memory. I will not show the whole surgery, you can it see later because it’s known for every neurosurgery how to do it. But I will go to the end of that film to show the post-operative MRI and what the patient will say three weeks after the surgery. This is the final stage where we’ve had some speech arrest for a couple of minutes, some memory disturbances, and we were very happy at this moment with that stage of resection of the temporal lobe. This is post-op MRI two days after surgery. The T1, the FLAIR, and the T2 sequences. And now, we’ll have a very short conversation with the patient.
Patient: [foreign language]
Dr. Tabakow: Okay. Thank you once again.
Nadja: That was a really, really interesting case. Thank you very much, Pawel. So, I would say, at this point, I would like to welcome all guest speakers back. So, you can turn on your cameras now. And we actually have one question to start out and you can also continue as a discussion so this can be very open, but one interesting question came from the audience, “How does edema affect the accuracy of the DTI information, and how do you,” maybe all three of you can answer how you do it, “consider this for you surgical planning?” Who would like to take it?
Puneet: Yeah, I’m happy to start. I think as we know, DTI is a…it’s an algorithmic program, which tells you the location of the fiber tracts but the fiber tracts do exist. Now, one of the criticisms of DTI has been that if you use different methods, you get different reconstructions of the tract. However, what is key is the spatial location of the tumor in relation to…the spatial location of the fiber tracts in relation to the tumor. Now, that doesn’t change, we looked at a very good…Einar showed a good DTI post-op, I think it was a cortical spinal, where you change these special settings and you could see the tract better. However, the spatial location has not changed, it doesn’t move to a different location. So, I think from a surgical perspective, it’s important to look at DTI and understand the spatial location of the tracts in relation to the tumor, whereas when we do awake surgery, I then look at that spatial location and find it with intraoperative subcortical stimulation.
Monosegmental tracts, tracts which carry one function, cortical spinal tracts and optic radiation have a very high sensitivity and specificity in location. Speech is a difficult one with subcortical because there is more than one tract in place. But even then, you’ve seen some good papers over the years, the sensitivity and specificity is very high. So, for us, just to finish up the question, it’s to understand the accuracy using subcortical stimulation for awake, and obviously, if you’re dealing neurophysiology for cortical spinal or optic radiation, then that’s a good way of looking at accuracy. So, thank you.
Nadja: Thank you for answering that one. Einar, Pawel, anything to add?
Dr. Vik-Mo: Yeah, I completely agree with Puneet and I think the main point here is, of course, what is our second option? I mean, it’s a technology we can use and we can get this idea where the fiber tracts are going. Sometimes, the edema is too dominant and you can’t really get any kind of information, but you know it’s only a supportive technology and I don’t think you can completely trust it but if you can find the fiber tract you’re looking for, that is stable.
Dr. Tabakow: I will also add to these great ideas of my colleagues that I showed in my presentation, a patient with bleeding from cavernoma, and see very, very visible edema that is fluid around this hematoma. And still, we could see the fasciculus arcuatus close to the tumor and pyramidal tract and they were indeed there. As Puneet said, the tracts don’t disappear. Maybe you can…if we have trouble before surgery in tract reconstruction, we go to the contralateral hemisphere and try to create a fiber, first of all, to know if we are good in this how, good we are in fiber construction. That is the easiest tip. Then, if we have a further problem, we go to our smart neuroradiologist and asked them if there is the destruction of this tract or just we cannot reconstruct.
And then they go to that mean diffusion ATC, they start to count the percentage of destruction of the fiber, and we do a very simple test with steroids, we increase the dosage of steroids. If the patient improves, for example, neurologically, that means that there is not direct destruction or injury, permanent injury of the tracts, so we can operate. And I will ask my colleagues about the non-invasive mapping of the brain, do you have experience with that module that enables…that has been shown to be very predictive? They’re using the transcranial or transcortical mapping of eloquent areas with the aim to decide if a patient has an operable tumor or not.
Dr. Vik-Mo: So, we don’t do mag evaluations in Oslo but, I mean, the technology is definitely there. On that topic, I think it’s difficult to say based on fMRI or DTI whether or not this is an operable case. I think in most cases, you would actually need to have the patient and do the monitoring to know for sure. And I think, also, if you can find the tracts and they fit with what you anatomically would expect them, then you could probably address it, but if you can’t find anything, then you don’t really know if it’s a dispersion of the tracts or if it’s technical or any other reason. So, I would be much more careful in saying that there is nothing there than I would say that the cortical tract probably is here, unless that’s where we’re going to go and look for it.
Puneet: Yeah, I don’t have an experience actually with transcranial magnetic stimulation. We’re at Oxford don’t have quite an expensive tool, mainly for cortical, this thing for motor, this thing for tracts. Personally, we don’t have it in Oxford. Yeah.
Dr. Vik-Mo: So, Puneet, could I ask you a short question regarding when you do the endoscopic approaches? So, then the idea would be to get into this region where you can push the tracts aside, so have you done post-operative imaging where you can actually show on the post-operative image that you have moved the tracts and then it moves back?
Puneet: Yeah, some of the scans I showed you with the endoscope-assisted approach, the post-op DTI, I showed you two or three cases of post-op DTI, and the idea there was, if you use the 30-degree endoscope, you’ll have 5-ALA fluorescence as well. Because you get a wider field of view, you can actually…in one of the cases I showed, you can actually go underneath the tracts and then take the tumor out. So, it’s possible and you can show it on post-op DTI. I think I showed three cases of post-op DTI where you can keep the tract intact.
Dr. Vik-Mo: Then you see the tract on the postoperative image like slightly dislocated and it moves back?
Puneet: Yes, yes, yes. So, once you’ve removed the tumor, that tract will never be in the same location, the post-op DTI will show the tracts in a new location. I don’t know whether we have any more questions in the chat, do we…?
Nadja: I think you did really well since you incorporated pretty much the questions that we had from the audience already in your answers. So, I think also considering the time, we would probably…or Emese, have you gotten any more questions in the chat that were not answered? I think we got it all covered.
Emese: I do think we got it all covered very well, I think we’ve seen very impressive and rich presentations here. So, if there’s nothing else to add right now, do you see my final slide?
Nadja: Not yet.
Emese: Not yet? Okay, let me show it real quick. Can you see it now?
Nadja: Yes.
Emese: Because I think, yeah, we’ve come to the end of this great webinar, it was very informative, and, yeah, we want to say thank you, first of all, to our three wonderful presenters, Puneet, Einar, and Pawel.
Nadja: A very warm thank you, yes.
Emese: Yes, we very much appreciate the time you took and, yeah, to share all the insights with us and especially with our attendees today. And also thank you to all the attendees this afternoon and for the questions you brought into this little discussion and Q&A. If any more questions come up or you need any more information, please contact us at this email, [email protected], and we are also happy to connect you with our three presenters if that’s needed. And, yeah, as mentioned, there’s going to be the recording of this session sent out to everyone who registered and will also be available as an on-demand video on brainlab.com. And I think with that, we wish you a great rest of the day. Most of all, stay safe and goodbye from us, and thank you.
Nadja: Thanks again.
Dr. Vik-Mo: Thank you.
Puneet: Thank you. Thanks, bye-bye.
Dr. Tabakow: Bye.
Emese: Thank you.
Related Webinars
Orthopedic Planning Mastery: Advanced Measurement Tools
Join us for an exciting webinar on advanced measurement tools in TraumaCad with Dr. Adam Rothenberg from EvergreenHealth (Kirkland, WA) on April 19th at 3:30 PM CEST. Gain valuable insights into your orthopedic planning workflow, including hip navigation planning, pre …
Adam Rothenberg, Dr. / MD
Knee Osteotomy Planning Webinar
Is Knee Osteotomy planning a topic of interest for you? If so, be sure to attend our upcoming webinar on Wednesday, July 27th at 6 pm UK time. We are excited to host in the session Mr. Raghbir Khakha, Consultant …
Raghbir Khakha, MD
Complex Joint Cases
Join us for an informative and engaging webinar on Thursday, May 12th at 7 pm CET / 8 pm IDT. The webinar will feature complex joint reconstruction cases that cover key details from the preoperative stage all the way to …
Alexander Greenberg, MD
See more upcoming webinars
Register now