Communiqués de presse
Brainlab Demonstrates Single-Use Blood-Repellent Markers for Software-Guided Surgery
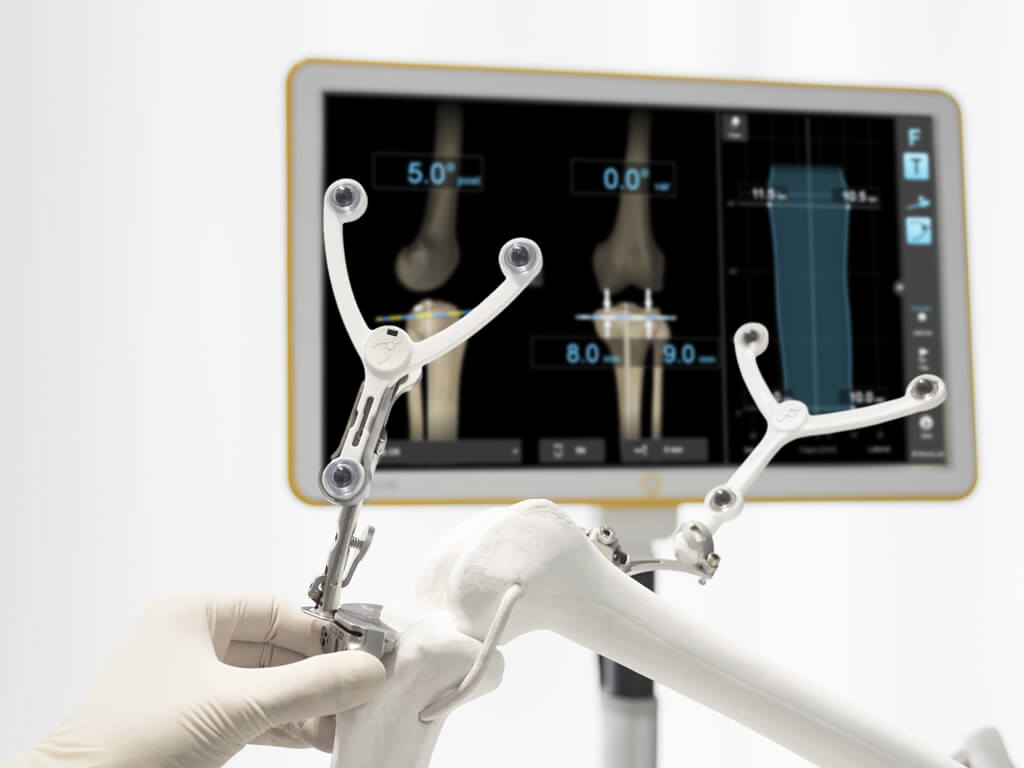
New technology feedback for ClearLens™ instruments garnered at American Academy of Orthopaedic Surgeons meeting
Orlando [AAOS], March 2, 2016—With a 2016 product launch in sight, Brainlab is seeking surgeon feedback on new technology at the American Academy of Orthopaedic Surgeons (AAOS) meeting, taking place this week in Orlando, Florida. ClearLens™* instruments are being designed for continuous and consistent tracking during software-guided surgery. Brainlab Software-Guided Orthopedic products allow physicians to plan, simulate and execute joint replacement procedures.ClearLens instruments are specifically designed to overcome known challenges in orthopedic navigation. The preassembled, single-use reference arrays snap quickly into place, remain visible to the surgical navigation system even when contaminated with tissue or fluids like blood and therefore may help to facilitate an undisturbed tracking performance and a smooth surgical workflow even in the most challenging clinical environments.
“Brainlab is continuously innovating in orthopedics to provide surgeons with software-guided tools, which may help reduce the risk of revision surgery while improving the overall functionality of the new joint,” said Martin Immerz, Vice President Orthopedics at Brainlab.
At the AAOS meeting, Brainlab will be presenting its full range of software-guided tools for orthopedics. In addition to garnering ClearLens feedback from attendees, Brainlab will also demonstrate Auto-Knee, automated knee templating which is part of the new TraumaCad 2.5 and visualization features for potential instabilities through the full range of motion, before bone cuts are made.
To discover the latest innovations in software-guided surgery from Brainlab, visit AAOS booth #2871 or brainlab.com/ortho.
* This product is not available for sale and will not be available until all applicable approvals have been acquired.
About Brainlab
Brainlab, headquartered in Munich, develops, manufactures and markets software-driven medical technology, enabling access to advanced, less invasive patient treatments.
Core products center on information-guided surgery, radiosurgery, precision radiation therapy, digital operating room integration, and information and knowledge exchange. Brainlab technology powers treatments in radiosurgery and radiotherapy as well as numerous surgical fields including neurosurgery, orthopedic, ENT, CMF, spine and trauma.
Privately held since its formation in Munich, Germany in 1989, Brainlab has more than 9,000 systems installed in about 100 countries. Brainlab employs 1,300 people in 19 offices worldwide, including 320 Research & Development engineers, who form a crucial part of the product development team.