Technology & the Treatment of Lung Cancer
Learn about lung cancer detection, current treatment options and what’s in store for the future of lung cancer therapy with radiotherapist-oncologist Professor Mark De Ridder, MD from UZ Brussels.
According to the American Cancer Society, lung cancer is by far the leading cause of cancer death. Each year, more people die of lung cancer than of colon, breast and prostate cancers combined [1].
We reached out to radiotherapist-oncologist Professor Mark de Ridder, MD, from UZ Brussels for his expertise and insights on lung cancer detection, current technology for lung cancer treatments and a look into the future.
Professor Mark De Ridder, MD graduated as a medical doctor from the Vrije Universiteit Brussel (VUB) in 1998 and obtained his accreditation as a radiotherapist-oncologist in 2005. He completed a PhD in medical sciences in 2005 and is full professor of oncology at the Faculty of Medicine and Pharmacy of the VUB. He has led the Radiotherapy Department and the Supportive Care Department at UZ Brussels since 2010.
In comparison to other forms of cancer, why is lung cancer so hard to detect at an early stage?
Dr. De Ridder: „Early-stage lung cancer remains asymptomatic for a long time. Patients often manifest symptoms when the cancer is at a more advanced stage because lungs do not have pain receptors and thereby do not cause noticeable discomfort till later on.“
What is the current standard of care for lung cancer?
Dr. De Ridder: „For node negative lung tumors smaller than 5 cm that are not invading other structures, like the main bronchi or vessels, the current standards of care are video-assisted thoracic surgery (VATS) or stereotactic body radiotherapy (SBRT).
Knives have been around since the time of the Greeks and Romans, thus making surgery the approach of choice for years. As such, surgery is still the standard of care for patients with those types of tumors that are in good physical shape.
However, many lung cancer patients may have comorbidities due to smoking or old age, thus making them non-ideal candidates for surgery. In contrast, these types of patients often receive SBRT. After receiving high doses of radiation, such patients can experience the same cure rate as they would after surgery – cure rates of up to 90%.
For the treatment of locoregionally advanced non-small-cell lung cancer, the volume of tumor and lymph nodes that needs to be irradiated is too large for SBRT. Therefore, the standard treatment changes and is more often a combination of chemotherapy and radiotherapy. Patients not progressing under radiotherapy still receive anti-PD-1 immunotherapy.
Patients with metastatic cancer would receive systemic therapy. There are also epidermal growth factor receptor targeting therapies and a greater variety of immunotherapies that can be combined with radiotherapy.“
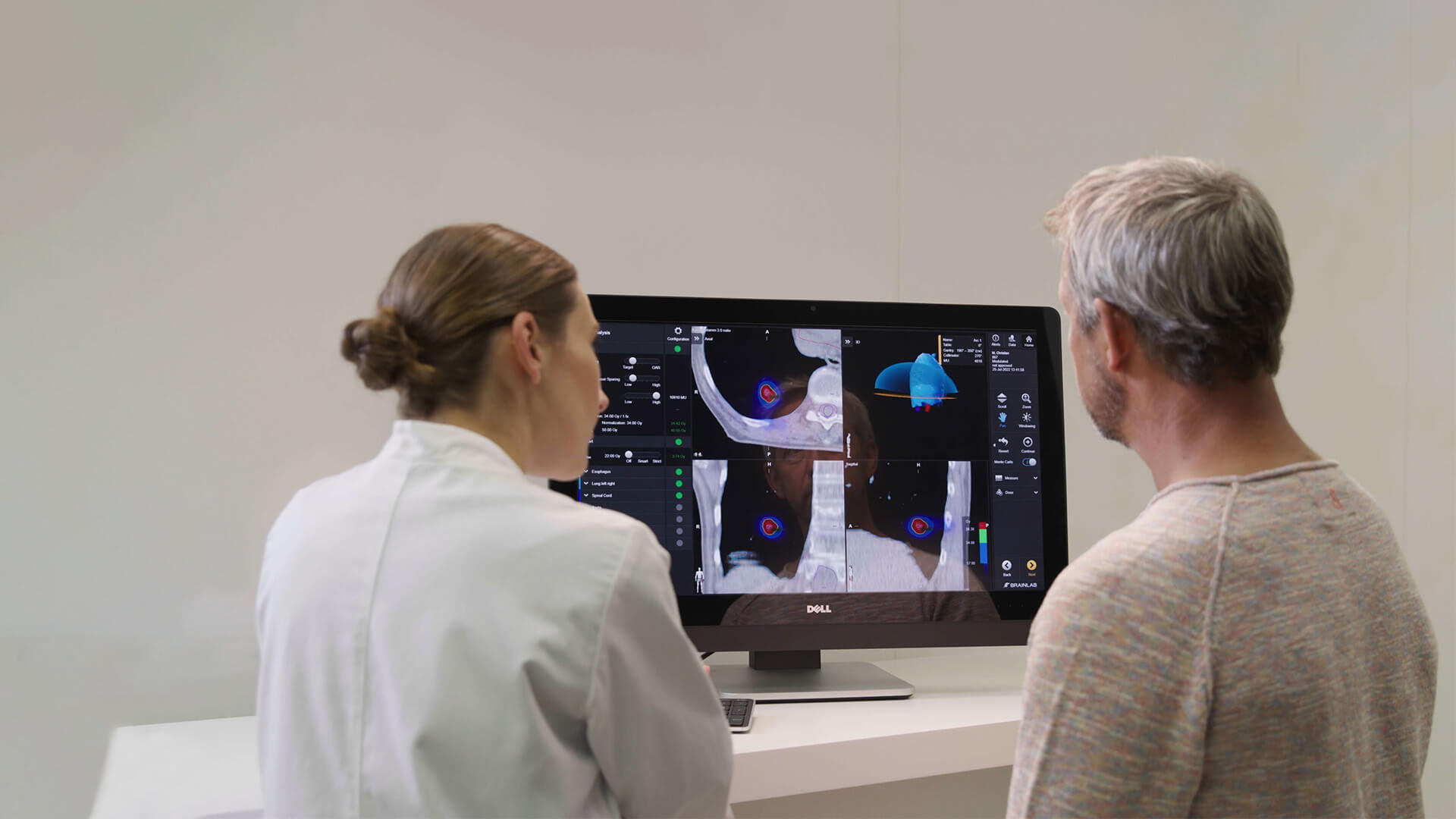
What is image-guided radiation therapy and surface-guided radiotherapy? Why are these two approaches relevant for the treatment of solid tumors and lung tumors?
Dr. De Ridder: „Image-guided radiotherapy (IGRT) means that you use imaging techniques to guide radiotherapy during the treatment phase. In the past, we used imaging during the planning phase, but over the last two decades, we’ve integrated the use of electronic portal imaging devices (EPIDs), cone-beam computed tomography (CBCT) and stereoscopic X-Rays. Ultimately, we use imaging to position and guide our therapy. However, one of the shortcomings of IGRT is that you don’t have continuous control during radiation.
In comparison to surface-guided radiotherapy (SGRT), you can take the surface of the patient into account to monitor their movement. For example, if a patient moves, you can hold the beam and then adapt your treatment table using IGRT and thereby avoid irradiating non-cancerous tissue.
In either case, you should have SGRT that is correlated to IGRT. This is what we do with our ExacTrac Dynamic® system. We have surface guidance, and when we see something is changing, we can correlate that with internal imaging using the stereoscopic X-Ray imaging or even by acquiring new CBCTs. The idea is to combine both approaches for precise and effective treatments.“
UZ Brussels has amassed years of valuable experience in treating patients with solid tumors, including patients with breast and lung cancer. How has the technology that UZ Brussels uses to treat lung cancer evolved?
Dr. De Ridder: „UZ Brussels has evolved considerably over the last ten years in terms of lung cancer treatment.
Many years ago, lung tumors were irradiated with large fields to compensate for the motion of the tumor during treatment. Because of the large volume of lung, esophagus and myelum that was irradiated, the treatment was toxic, and the dose could not be escalated till tumoricidal levels.
The natural next step was to make treatments more precise. For this we used our Novalis® system which required placing fiducial markers in patients and treating them by using gating – a treatment technique that guides the delivery of radiation as a person breathes and only delivers radiation when the tumor is in the treatment field. This enabled us to do dose escalation and thereby administer more doses of radiation to the tumor. However, the implantation of fiducial markers in lung cancer patients that may already have chronic obstructive diseases and emphysema can result in a 20-30% likelihood of pneumothorax (collapsed lung). This was most of the time easily remedied by aspiration, but it did not make for an elegant solution.
Then, in 2009, we became the first center outside of Japan to acquire the Vero® system. With this approach we could track the tumor in real time, without the additional treatment time imposed by gating. In any case, we were still working with fiducial markers. If the tumor moved more than 8 mm peak-to-peak amplitude, we implanted a marker into the tumor and used dynamic tumor tracking as a protocol for the patients. In contrast, if the tumor movement was limited, we would just perform a respiratory correlated computed tomography (4DCT) scan during the simulation step and irradiate the tumor with an internal target volume (ITV) technique with margins encompassing the entire movement. The big advantage of this approach was the increased efficiency of treatment.
With gating in general, you need to wait until the tumor is inside the gating window, whereas when using tumor tracking, you can administer radiation doses during the entire respiratory cycle. This increases the speed of treatment by a factor of 2 to 3, which of course improves quality of life for patients.
Unfortunately, at one point in time, we had flooding in our hospital and our Vero® system became unusable. As a result, we purchased an MRI-guided radiotherapy (MRgRT) system. So, the current standard of care at UZ Brussels is that we do 4DCT simulation, and if the peak-to-peak amplitude is limited, we treat the patient using an ITV concept with ExacTrac Dynamic® for surface guidance. If the tumor is moving too much and we can’t see the tumor on a classic linear accelerator (LINAC), we use MRgRT.
However, a big drawback of MRgRT is its price. Based on this, we decided to collaborate with Brainlab to develop markerless tumor tracking in ExacTrac Dynamic® to enable better motion management for LINACs. I think within the next one or two years we will be able to treat patients with 4DCT simulation, 4D-CBCT and markerless tumor tracking with integrated image and surface guidance using ExacTrac Dynamic®.“
How do you anticipate treatments for lung tumors and solid tumors will change over the next five to ten years?
Dr. De Ridder: „Over the last two decades, we had a revolution in radiotherapy techniques, and we are now able to administer radiation therapy with an accuracy of 2 mm for all body sites. As such, pushing the resolution of radiotherapy to less than 2 mm will not necessarily result in a major clinical benefit. Based on this, I believe that future treatments will be more focused on individualized patient dose prescription.
In specific, during the next five to ten years, we may be able to prescribe radiation therapy that is tailored to the patient. It’s not only about administering the right dose to the tumor, it’s also about integrating the biological signature of the tumor. To achieve this, radiotherapy vendors should collaborate with academic and research institutions to develop individualized patient treatment algorithms.
In addition, automatization of target delineation and treatment planning will further increase the quality of radiotherapy for lung cancer.“
1. American Cancer Society. Key Statistics for Lung Cancer. Lung Cancer Statistics | How Common is Lung Cancer?.